What are the ingredients of rubber and why does it rot?
What are the ingredients of tyre and why does it rot?
In the construction of rubber, various chemical, natural and mineral ores are used, each of which plays a special role in the performance of the tire as well as in its production. In the construction of rubber, various chemical, natural and mineral materials are used, each of which plays a special role in the performance of the tire as well as during its production.
The main ingredients of the tire are as follows:
Rubber
In terms of preparation and processing, rubber is divided into three categories:
- Natural rubber (Standard Malaysian Rubber)
- Synthetic rubber (Styrene butadiene rubber ,poly butadiene Rubber
- Reclaim Rubber
- Rubber process oil
Natural rubber is obtained from special sap grown in the tropics and in countries such as Malaysia and Thailand. One of the most famous brands of natural rubber is Standard Malaysian Rubber (SMR). Synthetic rubber is also an oil derivative and a product of petrochemical plants. Types of synthetic rubber include butadiene rubber (BR) and styrene butadiene rubber (SBR). Each of the different tire components contains different proportions of natural and synthetic rubber, depending on the physical properties required. Natural rubber, for example, has high thermal resistance and excellent adhesion, and is therefore more useful in the inner components of tires, such as layers.
Soot
Adding soot to the rubber increases the wear resistance of the tire, improves its mechanical properties. The structure of rubber is such that there are empty spaces between its molecules and soot is placed in these spaces. Sometimes non-soot fillers such as calcium carbonate or kaolin are sometimes used in addition to soot.
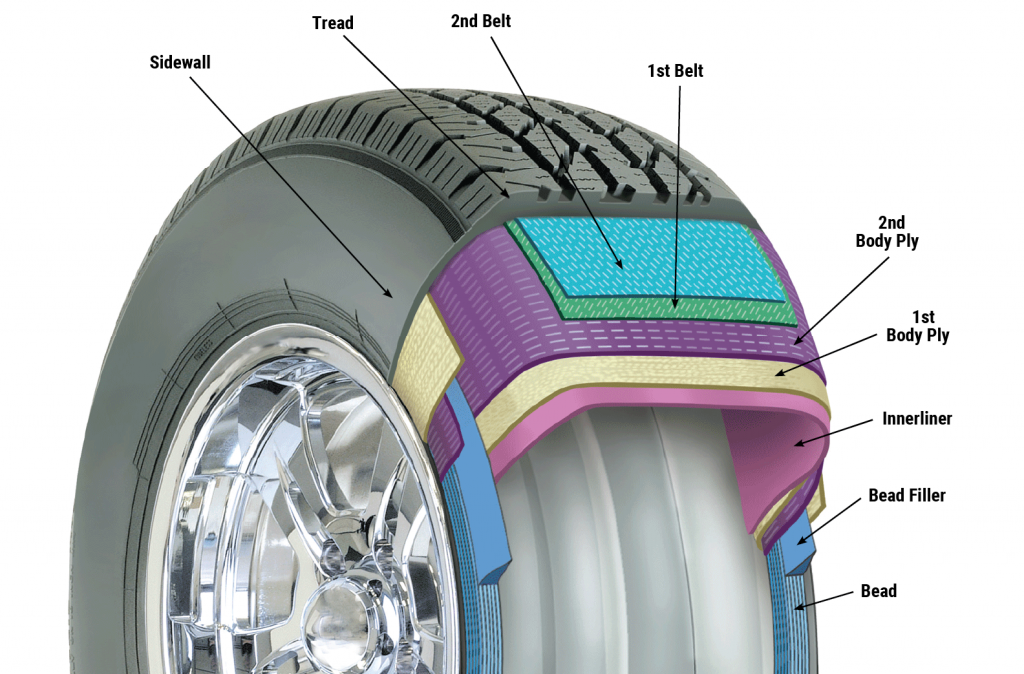
Thread
The main body of the tire consists of layers of cotton covered with a rubber compound (paste). The yarns are made of nylon, rayon or polyester. The sum of the layers of cotton used in a tire is called a carcass or carcass. The function of the cotton layers is to withstand the wind pressure from inside the tire and the load applied to the tire from outside.
Wire
In order to create sufficient strength in the part of the tire that is placed on the rim and is called the crown (willow), steel wires are used. In addition to having a copper or bronze coating, these wires are also covered by a rubber compound. After spinning for a few rounds, they form a set of crown wires with a square or trapezoidal cross-section. Also, in the belt of riding wire radial tires and in the layers of all-wire cargo tires, steel wires with a certain diameter and thickness are used.
Protectors
Oxygen and ozone in the air can damage and erode tire components. To counteract the harmful effects of these agents, special chemicals such as oxidants, waxes and anti-ozonants are used in the rubber compound.
Other chemical agents
In order to produce and process rubber mixes better and easier, in different cycles and stages of the production process from other materials such as oils, accelerators, sulfur (as a curing agent) and. . . is used.
Note: The amount of use of each of the above components depends on the type and structure of the tire, road traffic conditions, weather conditions and various other factors. For example, the weight percentage of each ingredient in a radial tire is as follows:
Natural and synthetic rubber. . . . 46-42%
Thread. . . . . . . . . . . . . . .. 5 – 3%
Soot. . . . . . . . . . . … . 26-22%
the wire . . . . . . . . . . . . . . . 15-11%
Other chemicals. . . . . . . 15-12%
Why is the car tire black?
Their blackness is due to the manufacturer’s efforts to increase the lifespan of their car products and increase its lifespan significantly. This paint is the result of a combination of stabilizing chemicals that are mixed with the tire polymer during production. This super chemical is called “Carbon black” and is added to protect your tire from UV damage and the ozone layer, and suddenly increases the life of the tire. Carbon black has the ability to resist the corrosion and gradual decay of ozone and absorbs ultraviolet rays to convert them into heat. By adding ozone to the molecules and chemical compounds that weaken the overall cohesion of the tire, the expansion and contraction of the tire will be less harmful and less hazardous, resulting in a longer and safer tire life.
The most important causes of tire damage are ultraviolet light and ozone.
Tires without these additives change color and disappear much faster. Unfortunately, these chemical adsorbents are consumed and lose their effectiveness over time. They are used to convert ultraviolet to heat and absorb ozone. When Carbon black loses its functionality, gray fades. This is one of the main reasons why black car tires wear out and turn a little white.
